Nucleophilic Acyl Substitutions of Anhydrides with Protic Nucleophiles Catalyzed by Amphoteric, Oxomolybdenum Species | The Journal of Organic Chemistry

PDF) Ester coupling reactions – an enduring challenge in the chemical synthesis of bioactive natural products
Efficient and Practical Procedure for the Esterification of the Free α-Car- boxylic Acid of Amino Acid Residues with β-(Trimet

PDF) Ester coupling reactions – an enduring challenge in the chemical synthesis of bioactive natural products
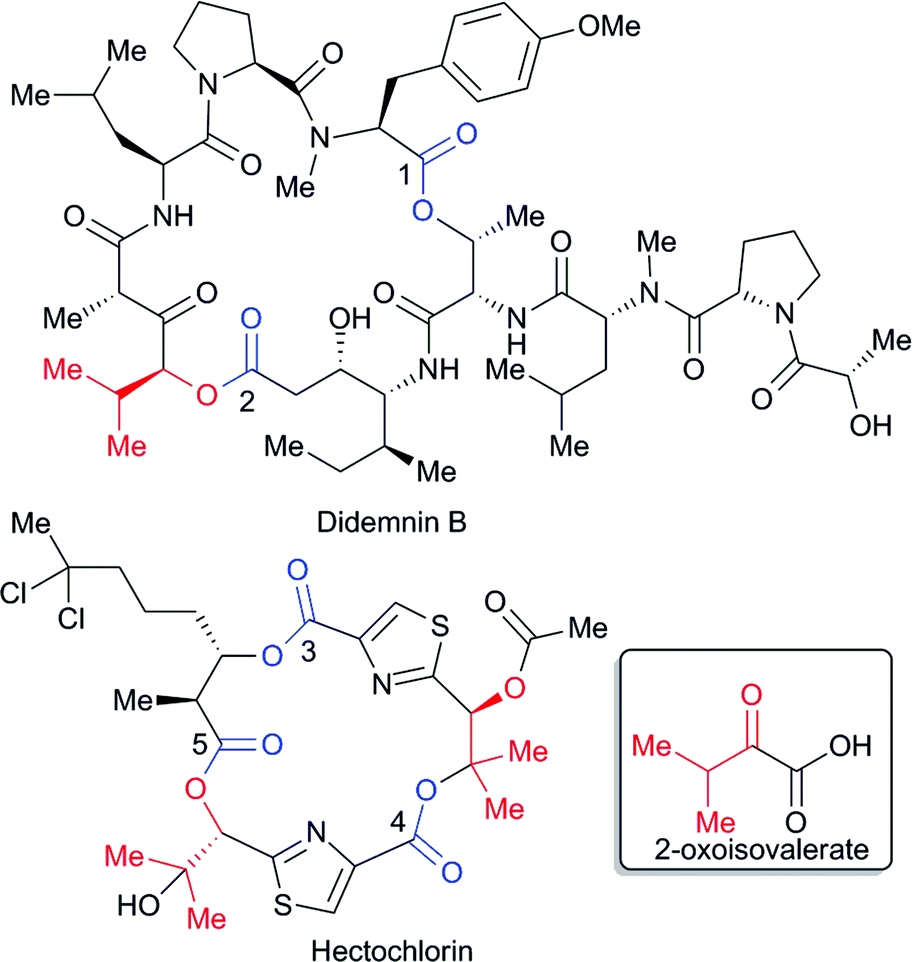
Ester coupling reactions – an enduring challenge in the chemical synthesis of bioactive natural products - Natural Product Reports (RSC Publishing) DOI:10.1039/C4NP00106K

Ester coupling reactions – an enduring challenge in the chemical synthesis of bioactive natural products - Natural Product Reports (RSC Publishing) DOI:10.1039/C4NP00106K

The ester derivatives obtained by C-ring modification of podophyllotoxin induce apoptosis and inhibited proliferation in PC-3M cells via down-regulation of PI3K/Akt signaling pathway - ScienceDirect

A fundamental developmental transition in Physcomitrium patens is regulated by evolutionarily conserved mechanisms - Jaeger - 2021 - Evolution & Development - Wiley Online Library

Synthesis, Evaluation, and Mechanism Study of Novel Indole-Chalcone Derivatives Exerting Effective Antitumor Activity Through Microtubule Destabilization in Vitro and in Vivo | Journal of Medicinal Chemistry
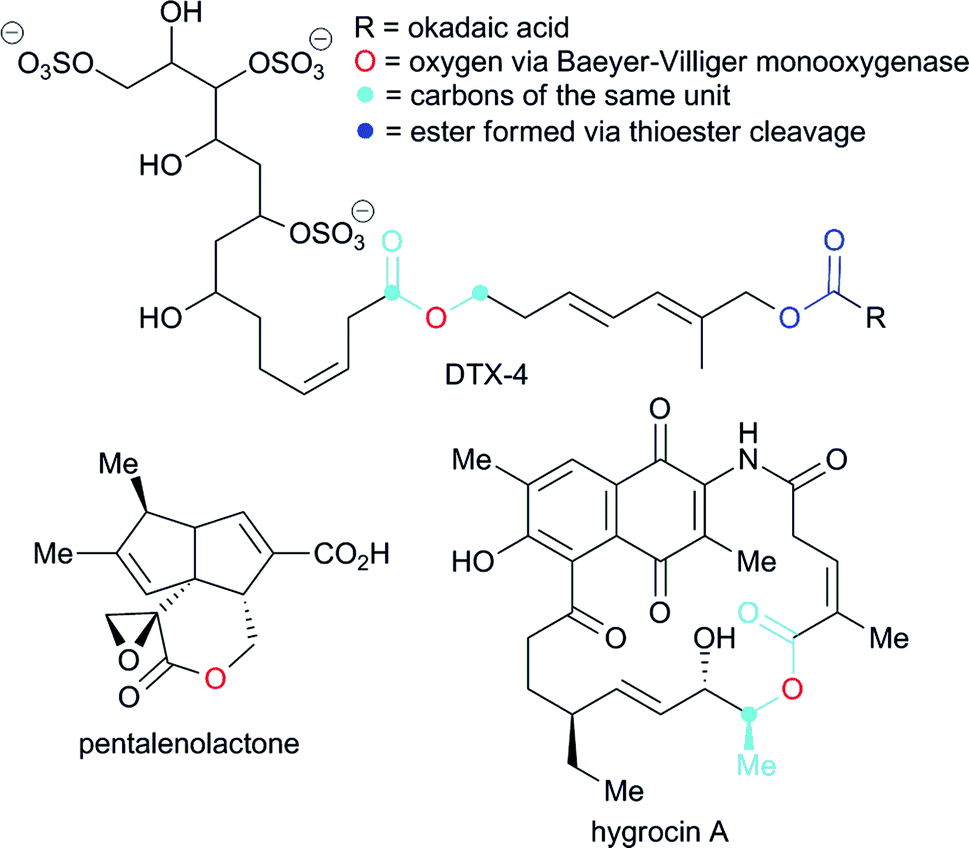
Ester coupling reactions – an enduring challenge in the chemical synthesis of bioactive natural products - Natural Product Reports (RSC Publishing) DOI:10.1039/C4NP00106K

Design, Synthesis, and Biological Evaluation of the First Podophyllotoxin Analogues as Potential Vascular‐Disrupting Agents - Labruère - 2010 - ChemMedChem - Wiley Online Library