SOLVED: In the reaction of nitrogen gas, N2, with hydrogen gas, H2, to form ammonia gas, NH3, how many moles of hydrogen are needed to react with 1.22 mol of nitrogen?
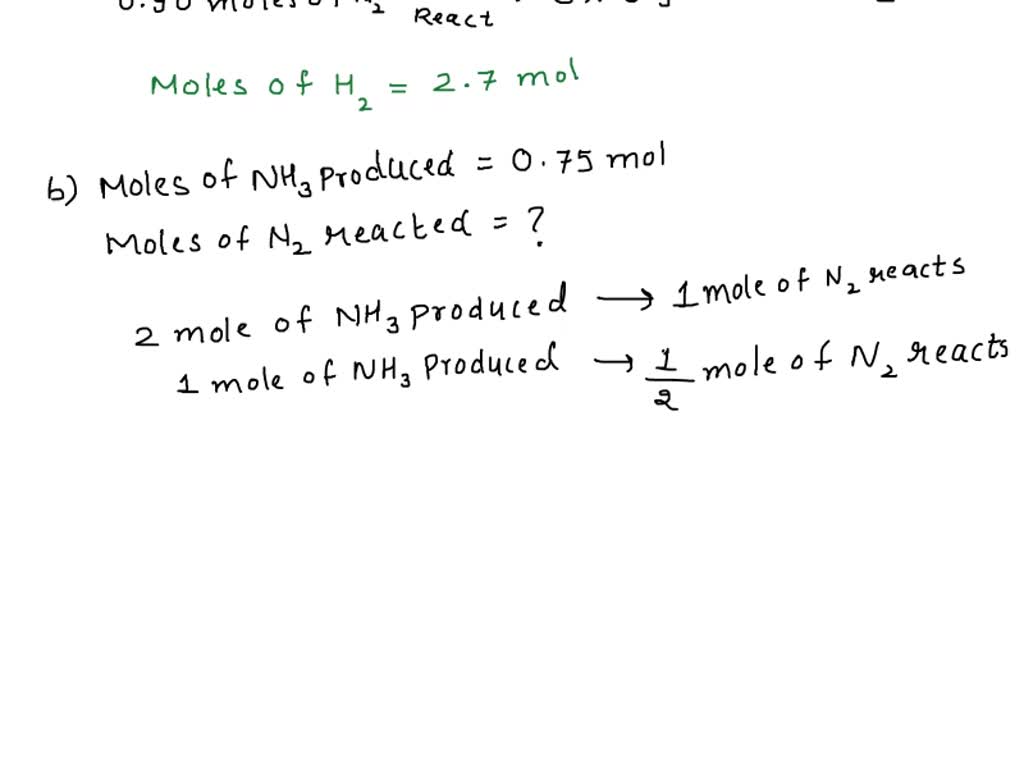
SOLVED: Ammonia is produced by the reaction of hydrogen and nitrogen as follows: N2(g)+3H2(g)→2NH3(g)ammonia A) How many moles of H2 are needed to react with 0.90 mol of N2? Express the number

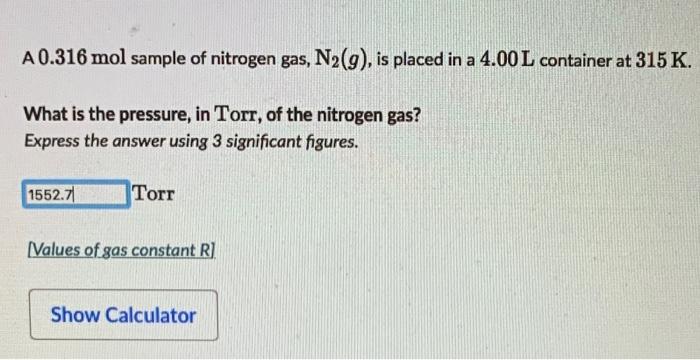
SOLVED: How many moles of N2 are in a 500.0 mL container at 780 mm Hg and 135 oC? options: 0.0153 mol 0.0146 mol 0.440 mol 0.463 mol