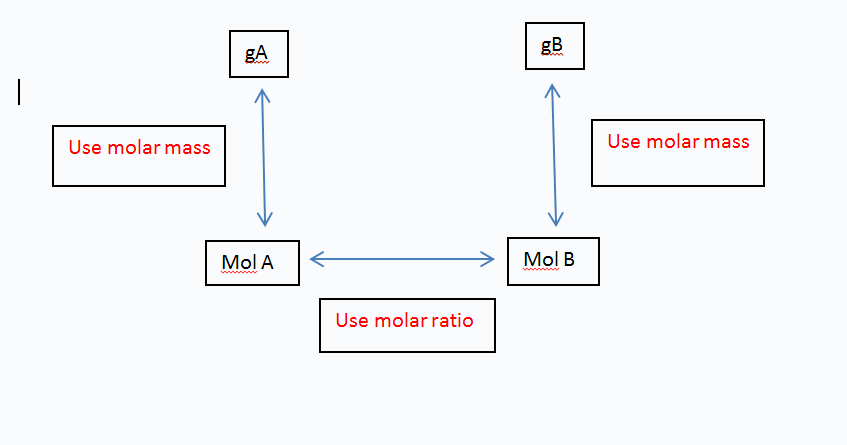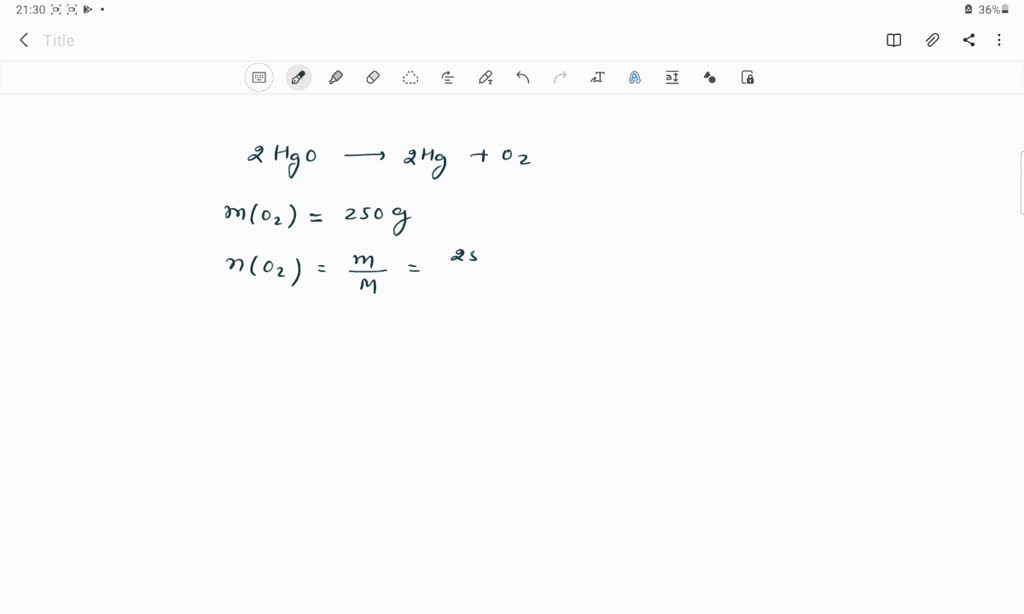
SOLVED: The molar mass of HgO is 216.59 g/mol. The molar mass of O2 is 32.00 g/mol. How many moles of HgO are needed to produce 250.0 g of O2?
1) At 2257 K and 1.00 atm total pressure, water is 1.77 per cent dissociated at equilibrium by way of the reaction 2 H2O(g) <

Question Video: Calculating the Mass of Calcium Chloride That Contains a Given Mass of Chlorine | Nagwa

In a reaction A + B2→AB2 , identify the limiting reagent, if any, in the following reaction mixtures.(i) 300 atoms of A + 200 molecules of B2 (ii) 2 mol A +