Amazon.com: Dr. Mercola H2 Molecular Hydrogen Dietary Supplement, 90 Servings (90 Tablets), Non GMO, Gluten Free, Soy Free : Health & Household

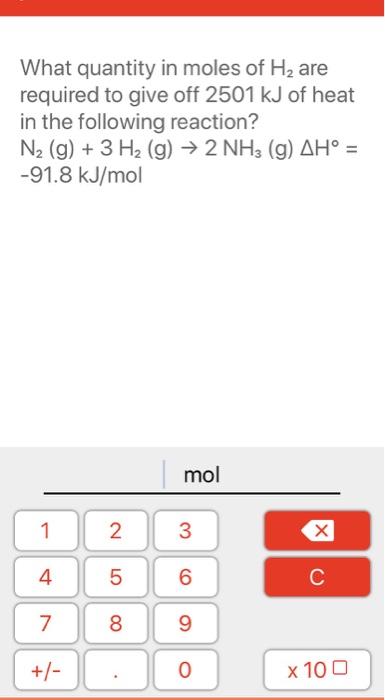
Welcome to Chem Zipper.com......: 1 mole of N2 and 3 moles of H2 are mixed in 8.21 lit. container at 300 K to form NH3 . If at equilibrium, average molecular mass

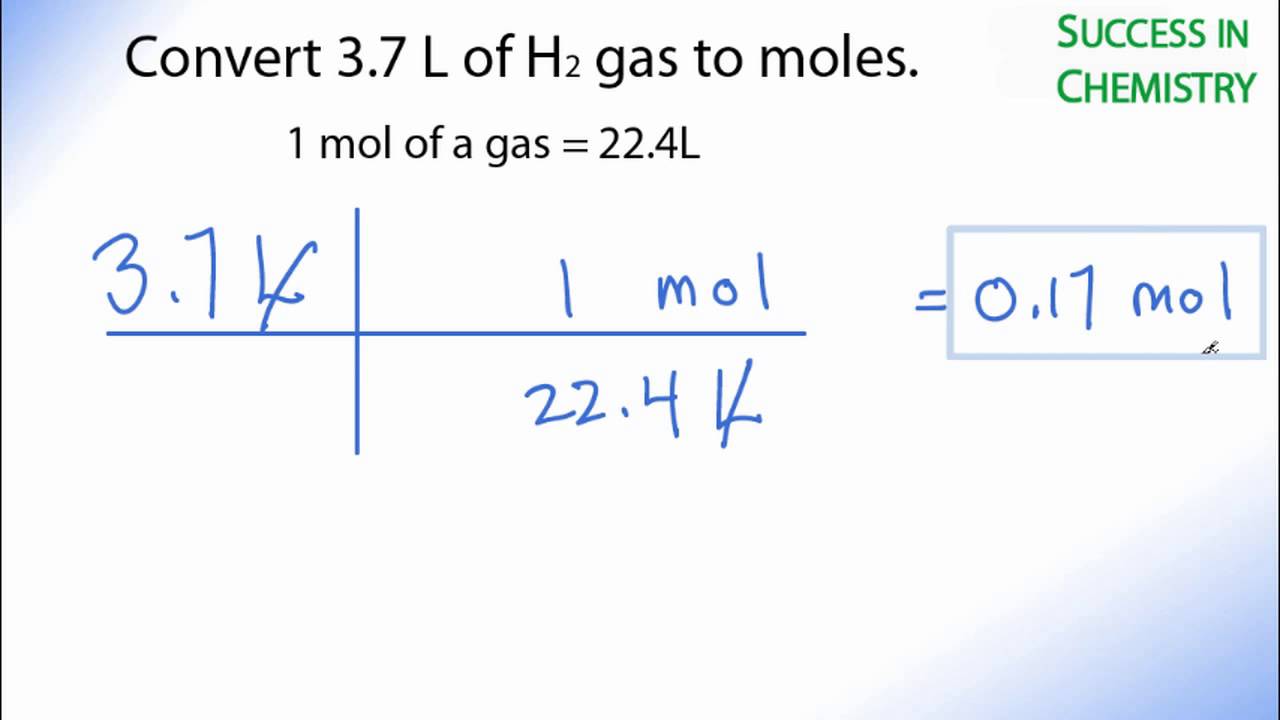
SOLVED: Which would have more molecules, 2.0 moles of hydrogen gas, H2, or 2.0 moles of methane gas, CH4? Explain your answer.

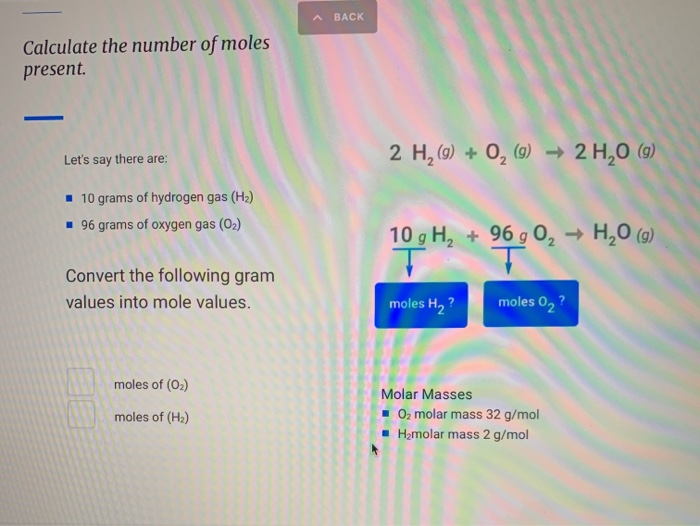
SOLVED: if 0.40 mol of h2 and .15 mol of o2 were to reat as completely as possible to produce h20, what mass of the reactant would remain