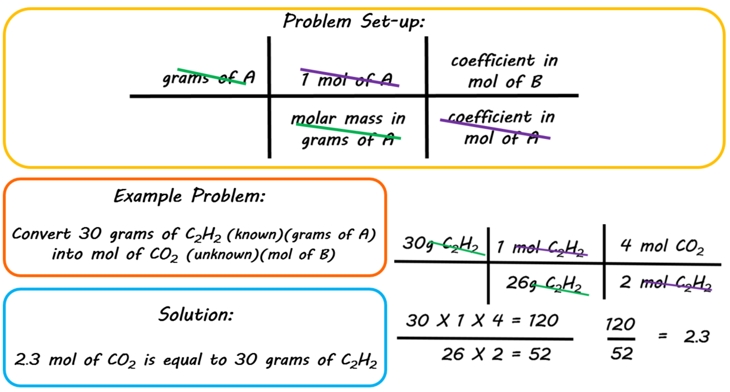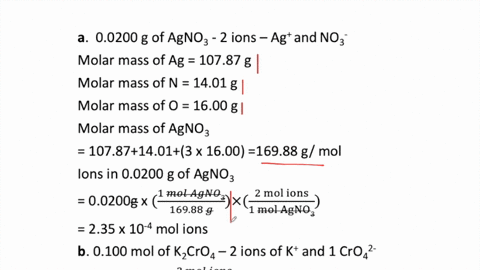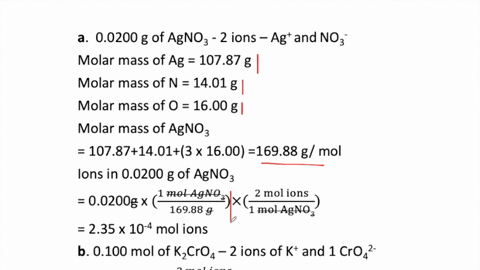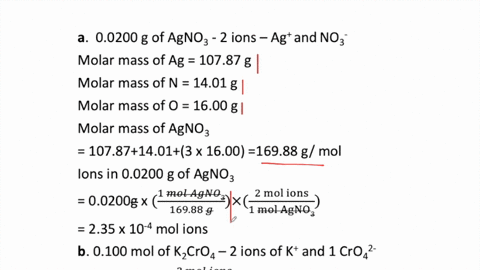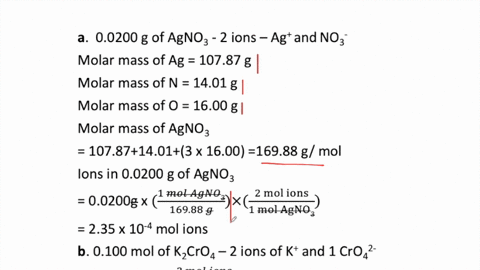
SOLVED:How many moles of ions are in each compound? a. 0.0200 g of AgNO 3 b. 0.100 mol of K2 CrO4 c. 0.500 g of Ba(OH)2 d. 1.00 ×10^-9 mol of Na2 CO3

For a chemical reaction 2X + Y → Z, the rate of appearance of Z is 0.05 mol L^-1 min^-1 . The rate of disappearance of X will be:
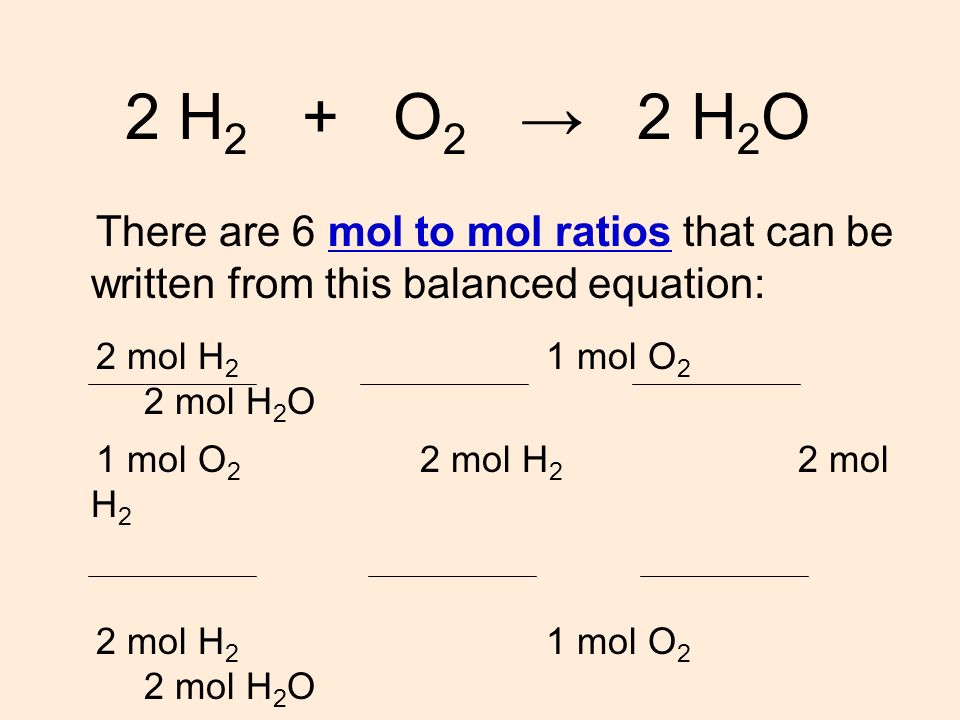
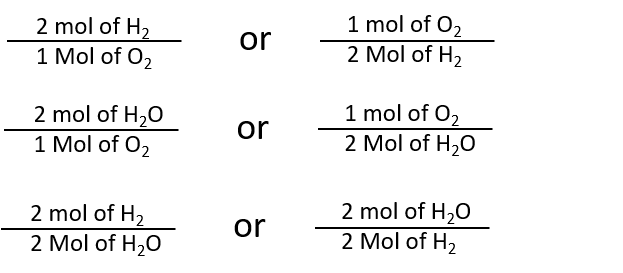
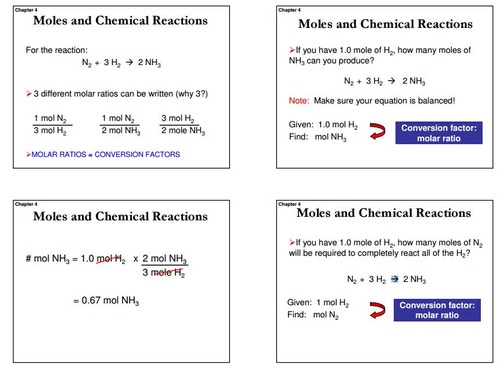
Mol ratio: coefficients of a balanced equation 2 H 2 + O 2 → 2 H 2 O 2 mol H 2 for every 1 mol O 2 In chemical calculations, mol ratios convert moles of. - ppt download

THE “MOLE” A Mole (mol) is a unit of measurement used for counting very small things (atoms, molecules, etc.) A mole of something is similar to a dozen. - ppt download