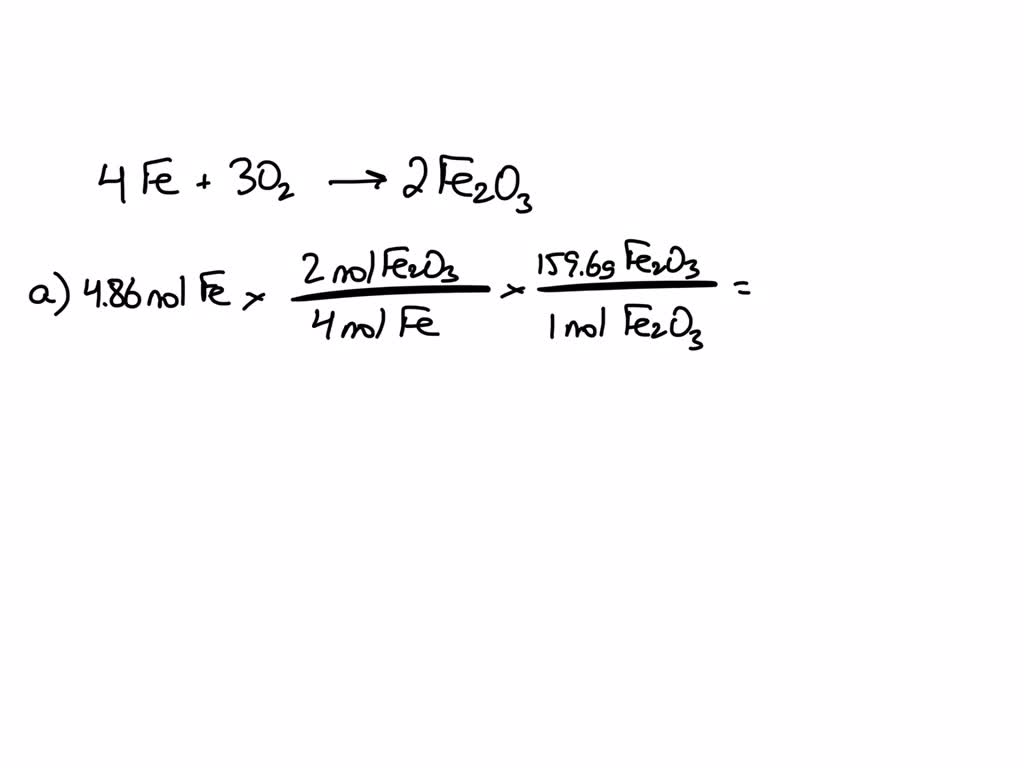
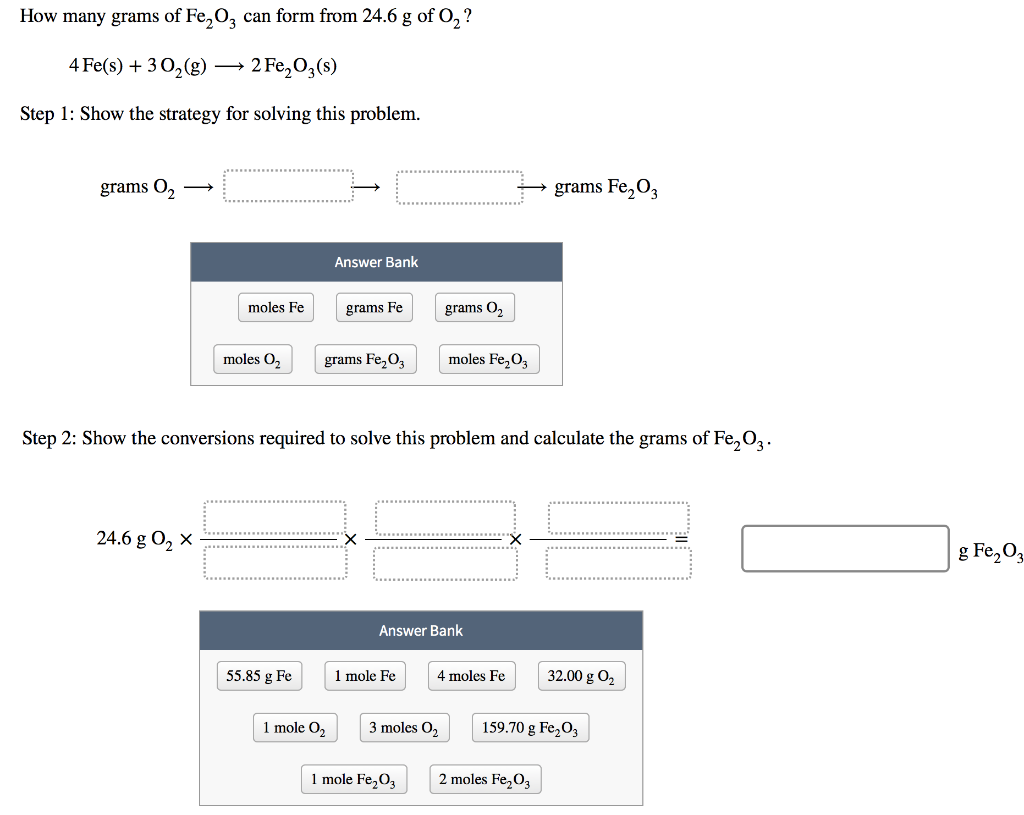
SOLVED: For the oxidation of iron to form iron(III) oxide: 4Fe(s) + 3O2(g) ⇌ 2Fe2O3(s) ΔS° = –549.73 J/K at 298 K The enthalpy of formation of Fe2O3(s) is –824.2 kJ/mol. What

SOLVED: Reaction: 2Fe2O3 + 3C –> 4Fe + 3CO2 1. Write the equation for this reaction. 2. If concentration of Fe2O3 is doubled, and concentration of C is tripled, what change in

4Fe + 3O2 = 2Fe2O3: How many grams of Fe2O3 will be produced from 10 g of Fe and 7 g of O2? - Brainly.com

SOLVED: Consider the following reaction: 4Fe (s) + 3O2 (g) —> 2Fe2O3 (s) If 5.0 moles of Fe and 4.0 moles of O2 are the present as the reactants, determine the limiting